Ácido acético ROTIPURAN® 100 %, p.a.
Bs. 32.80 – Bs. 231.15
Ácido acético glacial, Ácido metanocarboxílico, Ácido etanoico
Fórmula empírica C2H4O2
Masa molar (M) 60,05 g/mol
Densidad (D) 1,04 g/cm³
Punto de ebullición (pb) 117,9 °C
Punto de inflamación (flp) 39 °C
Punto de fusión (pf) 16,64 °C
ADR 8II
WGK 1
N° CAS 64-19-7
EG-Nr. 200-580-7
ONU-Nr. 2789
| Tenga en cuenta que este producto puede estar sujeto a sanciones legales como, por ejemplo, la obtención de una declaración de usuario final o la prohibición de algunas aplicaciones. Si desea más información, póngase en contacto con nuestro departamento de ventas. | |
 |
Ejemplos de efectos: Inflamable; los líquidos forman compuestos con el aire que pueden llegar a ser explosivos; producen gases inflamables con el agua o pueden autocombustionarse. Seguridad: Mantener alejado de llamas abiertas y fuentes de calor; cerrar bien los recipientes; almacenar de forma segura contra el fuego. |
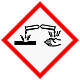 |
Ejemplos de efectos: Daña los metales y quema los tejidos corporales; puede causar lesiones oculares graves. Seguridad: Evitar el contacto; llevar gafas y guantes de seguridad. En caso de contacto con los ojos y la piel, aclarar con agua. |
| H226-H314 | Líquido y vapores inflamables, provoca quemaduras graves en la piel y lesiones oculares. |
| P210 P280 P301+P330+P331 P303+P361+P353 P305+P351+P338 P310 | Mantener alejado del calor, superficies calientes, chispas, llamas abiertas y otras fuentes de ignición. No fumar, llevar guantes/ropa de protección/protección ocular/protección facial, EN CASO DE INGESTIÓN: enjuagar la boca. NO provocar el vómito, EN CASO DE CONTACTO CON LA PIEL (o el cabello): Quitarse inmediatamente toda la ropa contaminada. Aclarar la piel con agua [o ducharse], EN CASO DE CONTACTO CON LOS OJOS: Aclarar cuidadosamente con agua durante varios minutos. Quitarse las lentes de contacto, si las hubiera y fuera fácil hacerlo. Continuar enjuagando, llamar inmediatamente a un CENTRO DE INFORMACIÓN TOXICOLÓGICA/médico. |
| Assay | ≥99.8 % |
| Colour (Hazen) | ≤5 |
| Aldehydes/acetaldehyde | ≤0.005 % |
| Sulphate (SO4) | ≤0.0002 % |
| Chloride (Cl) | ≤0.0001 % |
| Calcium (Ca) | ≤0.00001 % |
| Sodium (Na) | ≤0.00005 % |
| Potassium (K) | ≤0.00005 % |
| Mercury (Hg) | ≤0.000001 % |
| Aluminium (Al) | ≤0.000005 % |
| Cadmium (Cd) | ≤0.000001 % |
| Copper (Cu) | ≤0.000001 % |
| Iron (Fe) | ≤0.00001 % |
| Lead (Pb) | ≤0.000001 % |
| KMnO4 reducing substances | complies |
| Evaporation residue | ≤0.003 % |
| Solidification point | ≥16.35 °C |
| Note: Product may crystallise. It can be liquefied by heating in a water bath to max. 40 °C. | |
Description
Ácido acético ROTIPURAN® 100 %, p.a.
Determinación del valor de yodo
El valor de yodo es una medida de los ácidos grasos insaturados en los glicéridos. Cuantos más dobles enlaces olefínicos haya en la grasa, mayor será el valor de yodo.
Determinación del valor de peróxido
El índice de peróxido de una sustancia indica cuánto peróxido contiene. En una mezcla de ácido acético glacial y cloroformo, el peróxido oxida el yoduro en yodo elemental. La cantidad resultante de yodo se vuelve a valorar utilizando una solución de tiosulfato (donde se utiliza una solución de almidón como indicador). La solución en blanco probada en paralelo se utiliza para calcular los resultados.
Prueba de Barfoed:
La prueba de Barfoed se utiliza para distinguir monosacáridos de disacáridos, oligosacáridos y polisacáridos. Se añade el reactivo de Barfoed a la solución de azúcar, que luego se calienta durante unos minutos en un baño de agua. Los monosacáridos adquieren un color rojizo, mientras que los disacáridos sólo reaccionan después de haber sido hervidos durante un período más largo.
Ácidos y Bases para Análisis (p.a.)
¡Carl ROTH ofrece ácidos y bases adecuados para diferentes necesidades!
Alta pureza
Alta consistencia de lotes
Especificaciones detalladas
Buena relación calidad-precio
Estándares ROTI®Calipure para sus necesidades de calibración
Carl ROTH ofrece una amplia gama de estándares de calibración y control. Estos productos se fabrican a partir de material de partida de alta pureza y se prueban especialmente para cada aplicación. El correspondiente certificado de análisis específico del lote está disponible en línea. Cada serie de productos ofrece una excelente relación calidad-precio.